 Dr. Adam Summers is trained as an engineer and mathematician, he turned to biology to satisfy a deep need to interact with nature. His research applies simple physics and engineering principles to animals to understand the evolution of complex behaviors like feeding, movement and reproduction. The images shown here are the raw material that underlies his research. He is a professor at the University of Washington’s Friday Harbor Labs, author of more than 90 papers, and was the scientific consultant on Finding Nemo. He is obsessed with fishes and has recently come to realize that the tools and aims of science, art and poetry are more congruent than he had supposed. All the cleared and stained fishes can be seen at http://www.picturingscience.com (Editor’s note: Adam was also featured in Gizmodo this week)
Dr. Adam Summers is trained as an engineer and mathematician, he turned to biology to satisfy a deep need to interact with nature. His research applies simple physics and engineering principles to animals to understand the evolution of complex behaviors like feeding, movement and reproduction. The images shown here are the raw material that underlies his research. He is a professor at the University of Washington’s Friday Harbor Labs, author of more than 90 papers, and was the scientific consultant on Finding Nemo. He is obsessed with fishes and has recently come to realize that the tools and aims of science, art and poetry are more congruent than he had supposed. All the cleared and stained fishes can be seen at http://www.picturingscience.com (Editor’s note: Adam was also featured in Gizmodo this week)
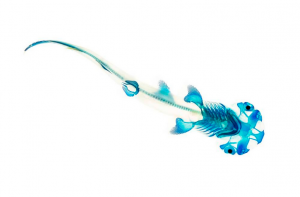
In my work I apply simple Newtonian physics and a bit of engineering to problems of how animals do those amazing things. The source of questions is the natural world and there is no better skill set in my business than that of the natural historian. A keen eye for what is going on around you, and a willingness to document it, is a powerful engine for generating great questions. Proposing answers to those questions almost always involves understanding the shape of the underlying system. So, the second most important set of tools I have are anatomical. Key insights are found in a deep understanding of the skeletal system and its associated soft tissue. A common tool in my lab is clearing and staining, which results in an entire specimen that has its mineralized tissue stained red and cartilage a deep blue.

This technique is not new. My own version is based on a version published 40+ years ago and refined by several authors since then (i.e. Dingerkus and Uhler ’77). This is certainly something that can be done in any lab, and even at home with some simple precautions. The raw material for this process is a dead vertebrate, ideally one that is less than 2 cm thick and fixed, fresh or freshly frozen. Feathers are a problem and so is fur, so if your vertebrate of choice has either you will have to remove the distraction. I pluck feathers and skin the fur bearers. Since this is a bit of a drag I would suggest starting with an amphibian or a ray finned fish. Staining the skeleton is a matter of exposing the specimen to tissue specific dyes. An acid/ethanol solution of Alcian blue will turn the cartilage and other proteoglycans blue. A soak in an aqueous solution with Alizarin Red S binds the red dye to the calcium phosphate in the skeleton.

Of course a stained skeleton is not of very much use if it can’t be seen, and that is where the ‘clearing’ comes in. Most of the unstained tissue is removed with a digesting enzyme called trypsin. This chews up many proteins but leaves collagen intact. This is important for the stability of the specimen, since the skin and connective tissue between skeletal elements will remain undamaged. A little high-test hydrogen peroxide takes care of any melanin in the skin or peritoneal cavity. At this point the specimen is white with an invisible but brightly stained skeleton. The last step is transparency. Collagen and glycerine have the same index of refraction, so by transferring the specimen into glycerine the skin, tendons and ligaments all become see-through.

The results are visually arresting but also very rich in anatomical information. Fine details of the skeleton can be seen without the perturbation that dissection entails. Radiographs are wonderful, but they are not 3-d and they won’t show cartilage. CT scans show mineralized tissue in 3-d, but they are expensive to generate and to process and again, they miss the cartilage. Magnetic resonance imaging will allow 3-D visualization of cartilage, but it is expensive and has lower resolution than I usually require. I suspect this classic technique will be fueling my research projects for many years to come.
Really interesting and beautiful photos. Interesting for from an artist’s point of view how I could use these different substances to create paintings.
Thats cool! 😀
Those are SO beautiful & precise, they make me lightheaded. Just amazing. Thanks for sharing!
They would be so much more beautiful had they still been alive and swimming instead of used as an art project.
They’re not “being used for art projects.” They’re being used for important scientific research. It just happens that the end result of that research is visually striking and has been popular on twitter recently.