Last week, Angelo spoke at thew 2024 Our Ocean Conference in Greece about going beyond 30×30 and how we equitably measure ocean success. You can also read his full prepared statement here: What I Meant to Say at Our Ocean Greece 2024
Canadian Masters students will now get up to $27,500 CAD a year, up from $17,500. Ph.D. students will get up to $40,000 CAD a year, up from as low as $20,000. Here’s how the leaders of Support Our Science did it. The following interview has been lightly edited for clarity and length. DS: Why was … Read More “Canadian grad students won their first raise in 20 years. Here’s how Support Our Science made it happen.” »
Dugongs and Seadragons is the only Dungeons and Dragons Podcast featuring actual marine science professionals playing D&D 5e while talking about marine science, conservation, and all things ocean. It’s been going for a long time, and jumping into the mid-point of an actual play podcast can feel like a daunting task. But we’ve got you … Read More “We Leased the Kraken! Catch up with the ongoing adventures of the Cephalosquad on Dugongs and Seadragons.” »
The great strength of 30×30, the national goal to protect 30 percent of the ocean by 2030, is that it sets out a framework wherein there is agreement that our shared use of public lands and waters fall along a spectrum, ranging from sacred places to wise use.
Our field is competitive, some job postings are confusing, and some career advice is contradictory or wrong. Here’s an exercise I have my students perform that I hope can help you. Graphic via Woods Hole Oceanographic Institution Hardly a day goes by that I don’t see a heartbreaking post from a prospective marine biologist in … Read More “Here’s what I teach my students about finding jobs in marine biology and conservation” »
Last month, I wrote a heartwarming little story about how doing a fun weekend hardware project with my daughter led to fixing one of the most annoying non-critical problems with the OpenCTD. After a month of testing, we have fully implemented the new power management system into the next iteration of the OpenCTD. Currently, there … Read More “Charging the OpenCTD is annoying, so we fixed it.” »
After a trio of very widely read articles triggered a traffic surge in February, including David’s critical expert analysis of cross-order hybridization, our visitor count normalized a bit on the old ocean science and conservation blog. A little more than 19,500 people visited Southern Fried Science in March, a roughly 50% increase from January. You … Read More “Space Crabs, Big Boats, and Fake Sharks: What you read on Southern Fried Science in March” »
A new study shows how unique cells full of crystals give this ray one of the brightest blue colors in nature Photo of Dr. Shahrouz Amini, MPI Potsdam, photographing a ray’s blue spots Coral reef fishes come in all the colors of the rainbow (and perhaps even in some colors that we can’t see). But … Read More “How the Bluespotted Ribbontail Stingray got its Spots” »
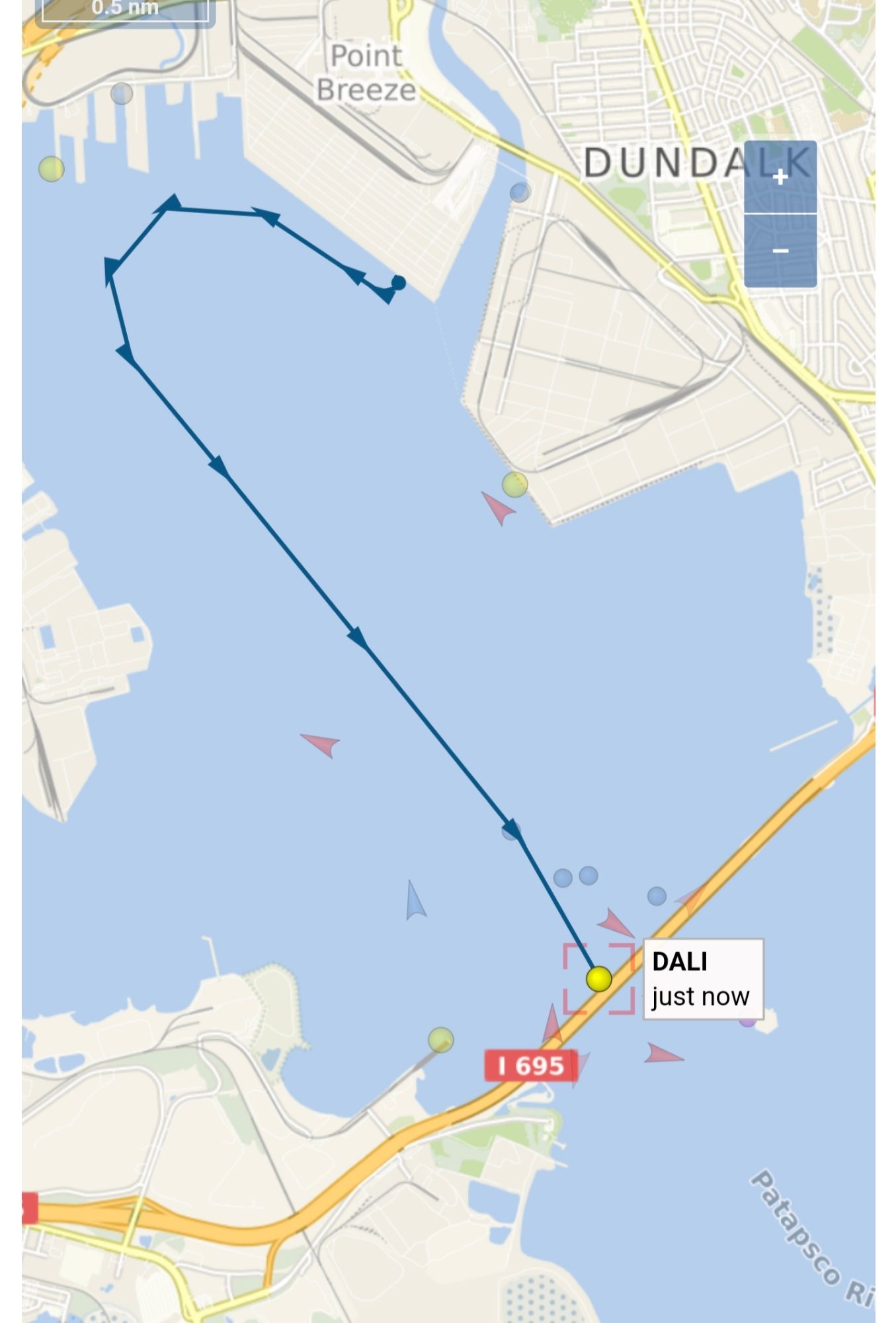
Early this morning, the cargo ship MV Dali collided with the Francis Scott Key Bridge, causing the bridge to collapse, sending several vehicles and people into the water. Search and rescue is currently underway. Because Twitter is now a clearinghouse for the worst and most disingenuous hacks on the web, there’s of course a rumor … Read More “No, the ship didn’t steer towards the pylon: A brief fact check on the MV Dali collision with Baltimore’s Key Bridge” »
The International Seabed Authority is meeting this month in Jamaica, but it is not the entire International Seabed Authority. Only the Legal and Technical Commission and the Council meet this months. The Legal and Technical Commission is a body of experts that reviews documents and proposals, usually in private as many contain privileged information from … Read More “What I’m watching for at this month’s ISA meeting: The Vibes” »